Induction of haploid zebrafish and analysis of resultant developmental defects and aberrant gene expression
FAYETTEVILLE, GA, UNITED STATES, April 14, 2026 /EINPresswire.com/ -- Researchers induced haploid zebrafish using UV-treated sperm and tracked why the embryos developed severe defects. They found that haploid embryos had low gastrulation and hatching rates, widespread malformations, and major changes in gene activity. The study points to disrupted FGF/Wnt signaling, mitochondrial function, ion transport, and cell-cycle control as key drivers of the lethal “haploid syndrome.”
Naturally occurring haploids are predominantly documented in plants; the first report dates to 1923, when haploid individuals were identified in the Jimson Weed. Haploidy also occurs in certain animal lineages, most notably in the haplodiploid sex-determination system of hymenopteran and dipteran, such as bees where males develop from unfertilized eggs and are therefore functionally haploid. However, experimentally induced haploid animals—particularly vertebrates—typically exhibit severe developmental defects collectively termed “haploid syndrome”, which culminate in embryonic lethality.
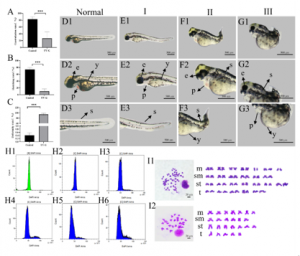
A new study published in the KeAi journal Reproduction and Breeding sheds light on why haploid fish embryos often fail to survive normal development. Researchers from Hunan Normal University created haploid zebrafish embryos by activating eggs with ultraviolet-treated sperm. Unlike normal embryos, which inherit two sets of chromosomes, these embryos carried only one.
“We found that haploid embryos appeared normal during the very earliest stages of development. However, as development progressed, serious problems began to emerge,” shares senior and co-corresponding author Liangyue Peng. “Many embryos developed curved body axes, swelling around the heart, reduced swimming ability, and died early.”
Notably, their rates of gastrulation and hatching were also far lower than those of normal diploid embryos. To investigate the cause, the researchers compared gene activity in three groups: haploid embryos, normal diploid embryos, and malformed diploid embryos.
“Using RNA sequencing, we identified 2,247 genes with altered activity, including 13 key genes that were consistently associated with abnormal development,” adds Peng. “Further analysis showed that these genes are involved in several essential biological processes, including developmental signaling, energy production, ion balance, and cell growth.”
In particular, the study pointed to disruptions in the FGF and Wnt signaling pathways, mitochondrial translation, ion transport, and cell-cycle regulation. Also, haploid syndrome does not appear to result from a single defect. Instead, it is likely caused by the combined breakdown of multiple developmental and metabolic systems that embryos need in order to grow normally.
“The findings help explain why haploid embryos, although useful for rapid genetic screening because recessive traits can be seen directly, are difficult to use in breeding and developmental research,” says Peng.
References
DOI
10.1016/j.repbre.2025.12.004
Original Source URL
https://doi.org/10.1016/j.repbre.2025.12.004
Funding Information
This work was supported by the National Natural Science Foundation of China (32072959) and Hunan Province Postgraduate Scientific Research Innovation Project (CX20250783).
Lucy Wang
BioDesign Research
email us here
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.